AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Cathode reaction in electrolytsis of cucl212/10/2023 To lower the melting temperature, calcium chloride is added to the sodiumĬhloride in a 1:2(NaCl:CaCl 2) part mixture. Issue of viability important but at this temperature molten sodium formsĪ thick fog inside the reaction vessel which is difficult to recover. Requires a significant amount of energy to be supplied. Now sodium chloride melts at approximately 800☌ this The highĬurrent passing through the cell provides sufficient heat to keep the The reaction mechanism of electrolytic reduction of SiO2 at a liquid Zn cathode in molten CaCl2 was investigated with the aim of establishing a new production process of solar-grade Si. Molten mixture and is collected in a water tight storage vessel. The reaction at the cathode: The Copper ions Cu 2 + have the lowest reduction potential. An iron mesh screen is used to preventĬhlorine gas, formed at the anode, from coming into contact with sodium Cu 2 + ( aq ) + 2 Cl - ( aq ) Cu ( s ) + Cl 2 ( g ). The cell consists of a central carbon anode surroundedīy a cylindrical iron cathode. The sea is full of it and we can easily get to it. Niether sodium nor chlorine, however, occur naturally in nature, but we do have a plentyfull source in the form of salt (NaCl). Write the reduction half-reactions that turn the neutral element into its ion by adding electron(s). Cyclic voltammetry and potentiostatic electrolysis were used to study the electro-reduction of the Cr 2 O 3-loaded metallic cavity electrode. Summary: Determine what ions came from the compound. 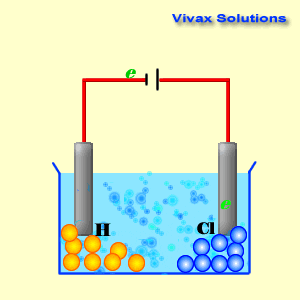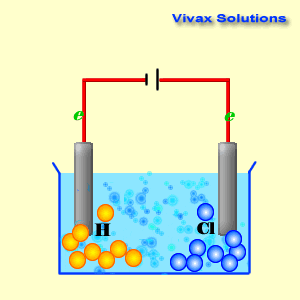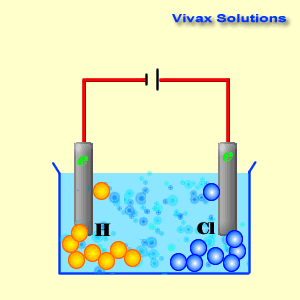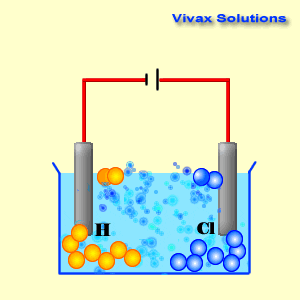
Sodium metal and chlorine gas are starting materials for the inudstrial production of a range of substances ranging from plastics to sodium fluoride found in toothpaste. The electro-reduction of chromium oxide (Cr 2 O 3) was investigated in an equimolar mixture of CaCl 2 NaCl molten salt at 800 ☌ for developing a more efficient process for chromium preparation. Consider the electrolysis of 1M CuCl2 at neutral pH.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
